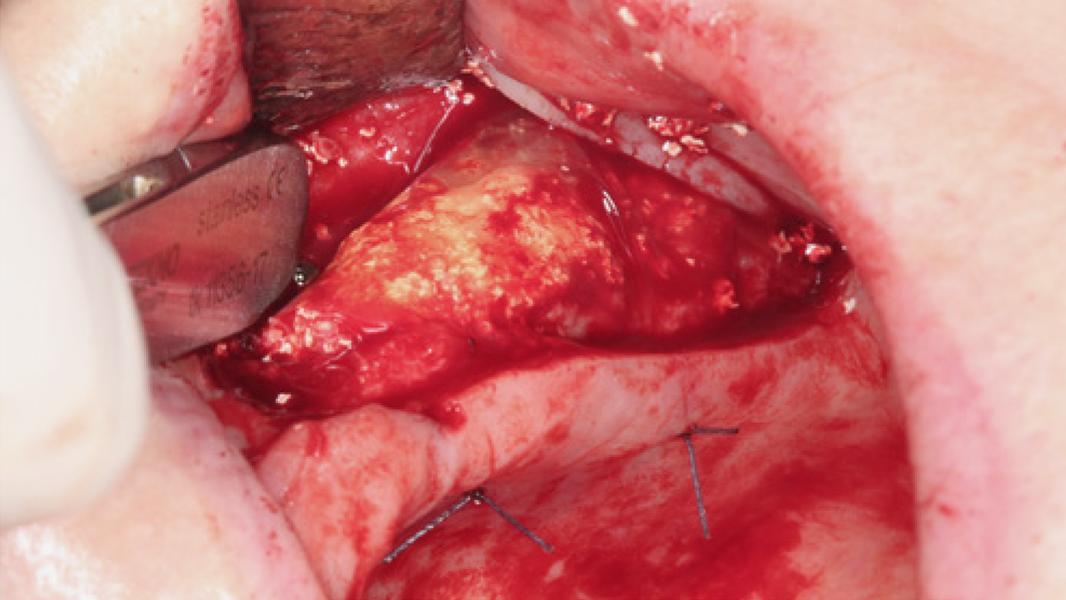
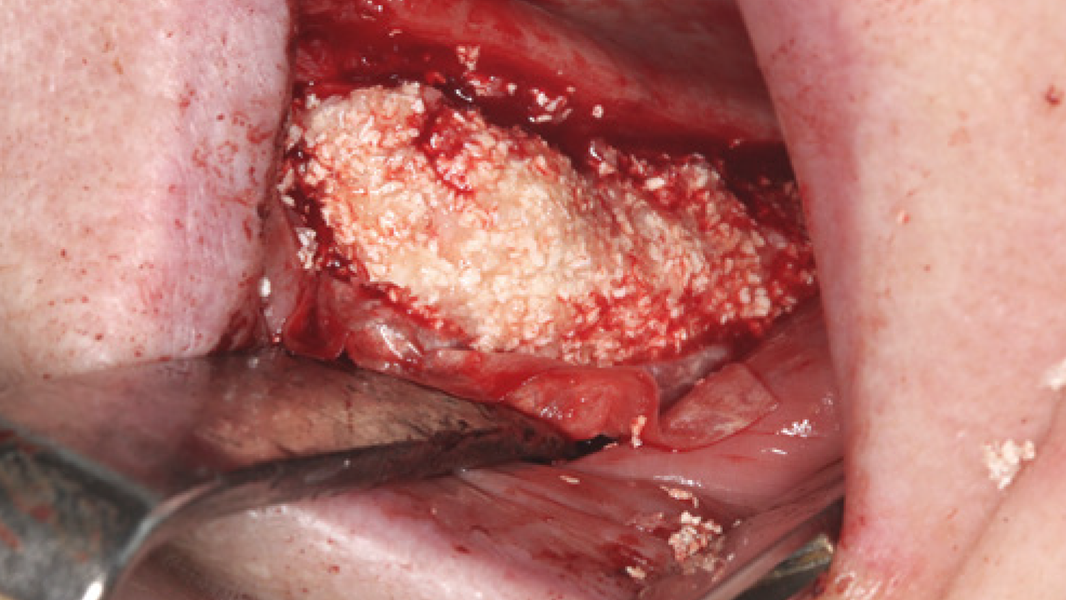
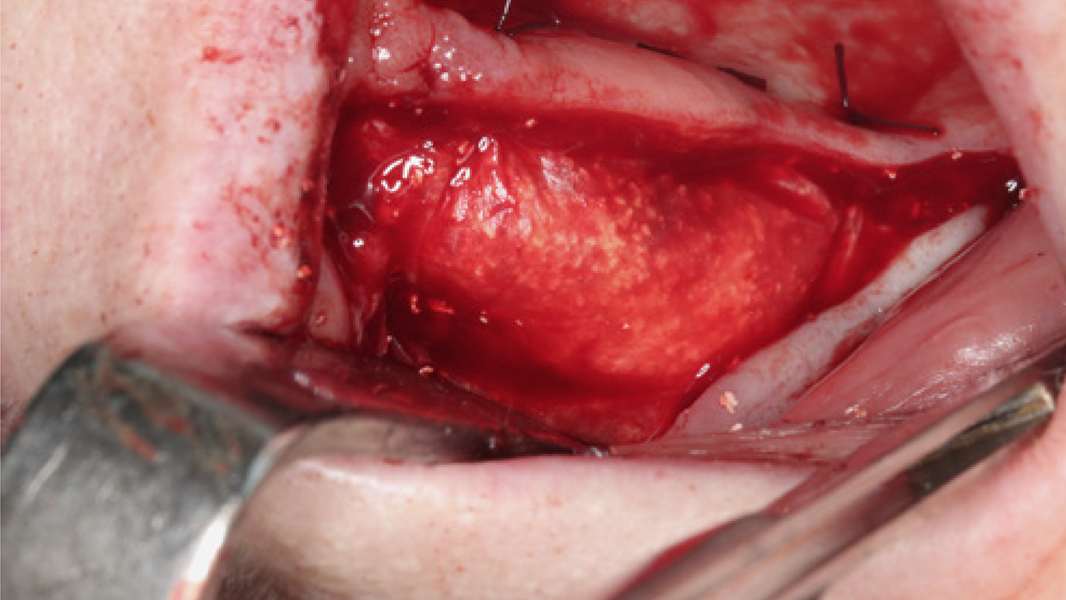
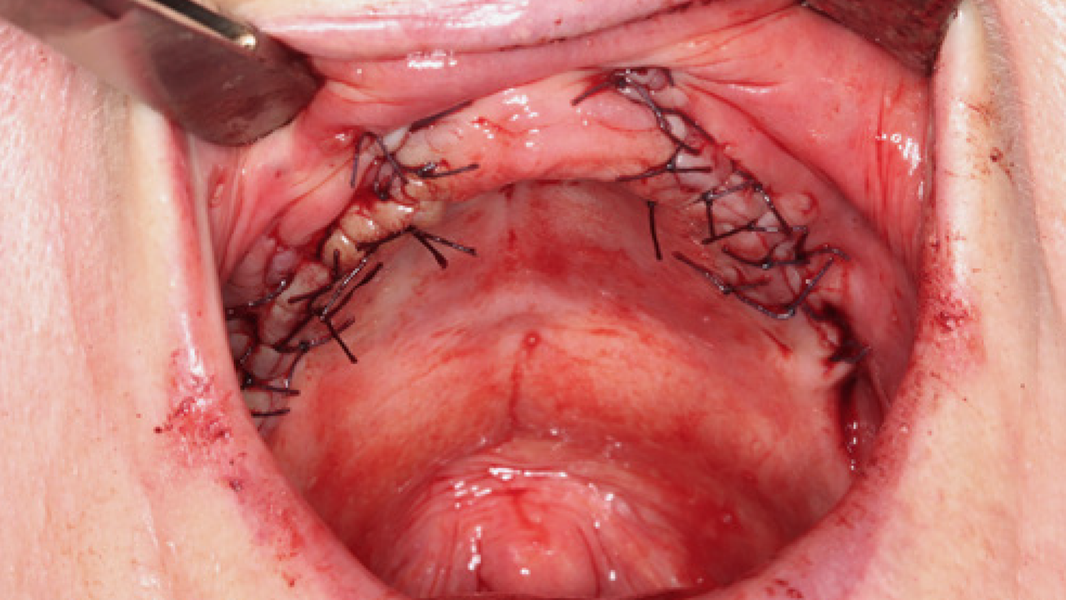
MinerOss® A Family
The alternative to autologous bone
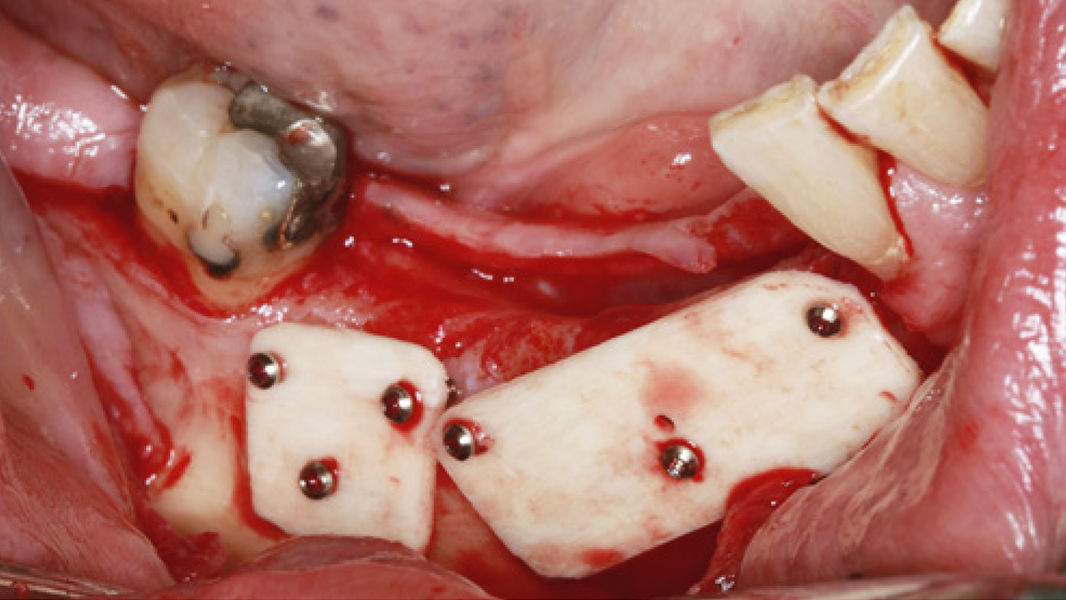
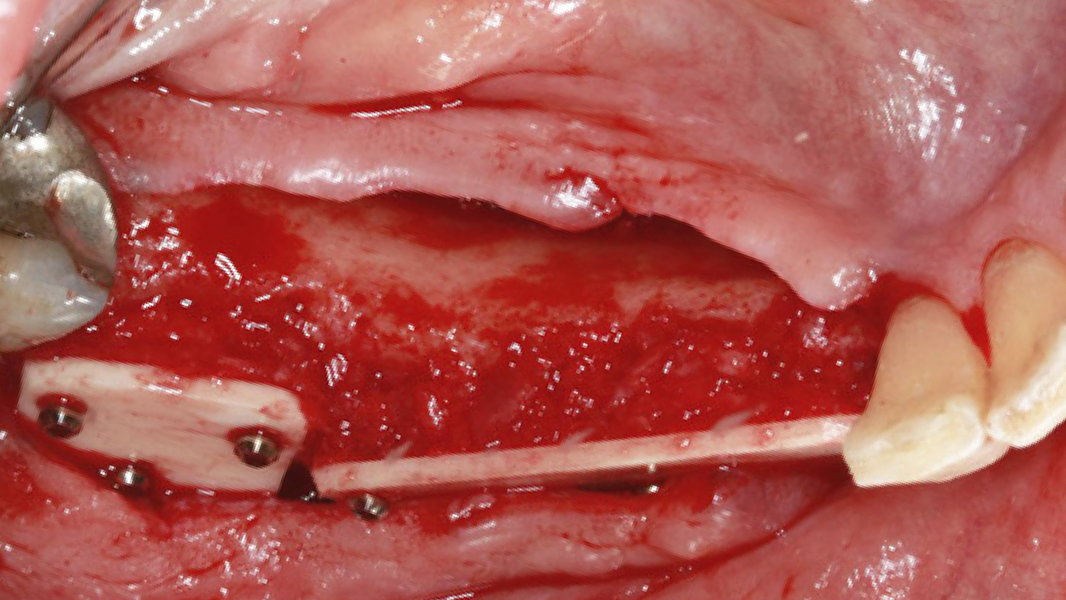
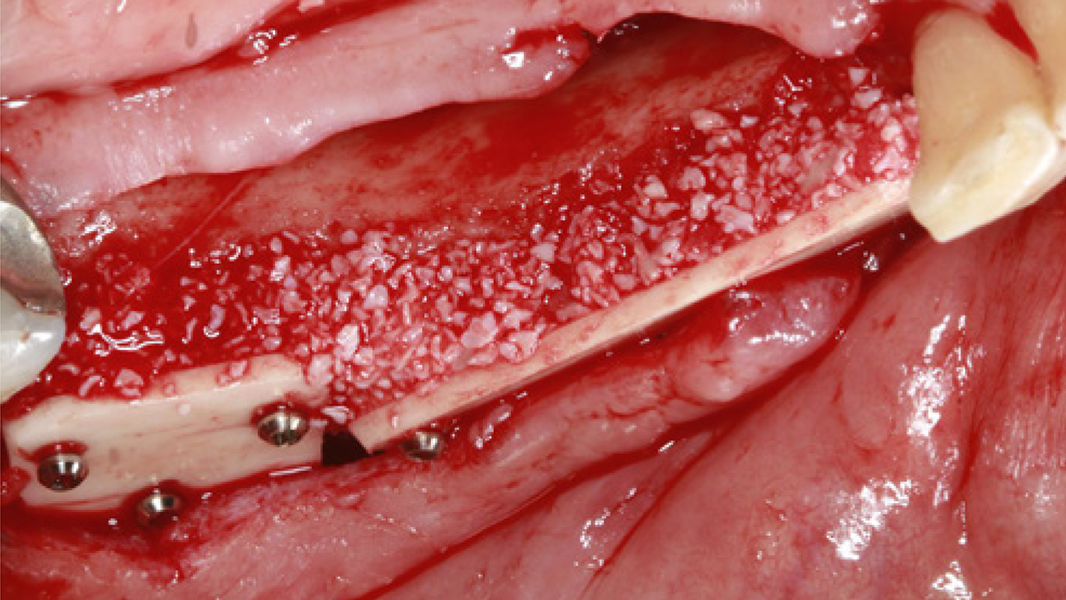
MinerOss A is a human allograft primarily derived from the femoral heads of patients undergoing hip replacement surgery. The donated tissues are thoroughly inspected and are subject to strict serological screening at the Cells+Tissuebank Austria (C+TBA). The Allotec process is used to clean and sterilize the tissues, yielding MinerOss A as cancellous or cortico-cancellous particulate, blocks, or cortical plates. A new formulation of granules mixed with sodium hyaluronate (hereinafter “MinerOss A + HYA”) is also available.
The cancellous variant supports revascularization and the supply of vital cells, thus promoting rapid regeneration. The cortico-cancellous formulation ensures higher volume stability when reconstructing extensive bone defects.
Studies demonstrated that allografts are comparable to autologous bone in terms of regenerative potential and physiological remodeling, unlike bone substitutes of xenogeneic or synthetic origin.1–5
Since the proprietary Allotec process preserves the native collagen in the material,6, 7 MinerOss A exhibits high regenerative capacity combined with a natural remodeling behavior.4
MinerOss A is therefore a predictable alternative to autologous bone grafting.
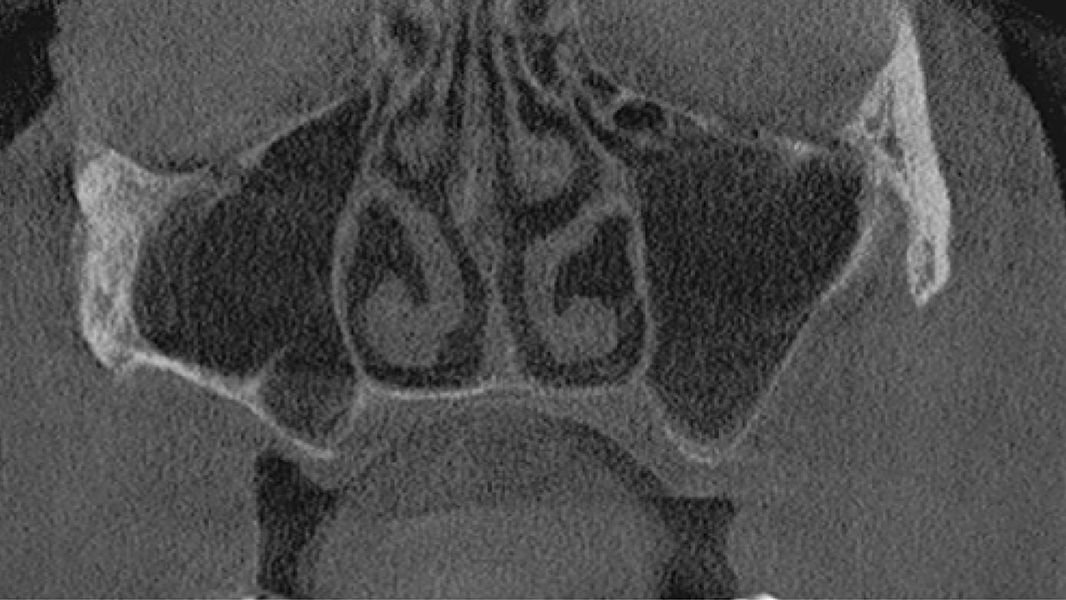
Features of the human bone substitute material
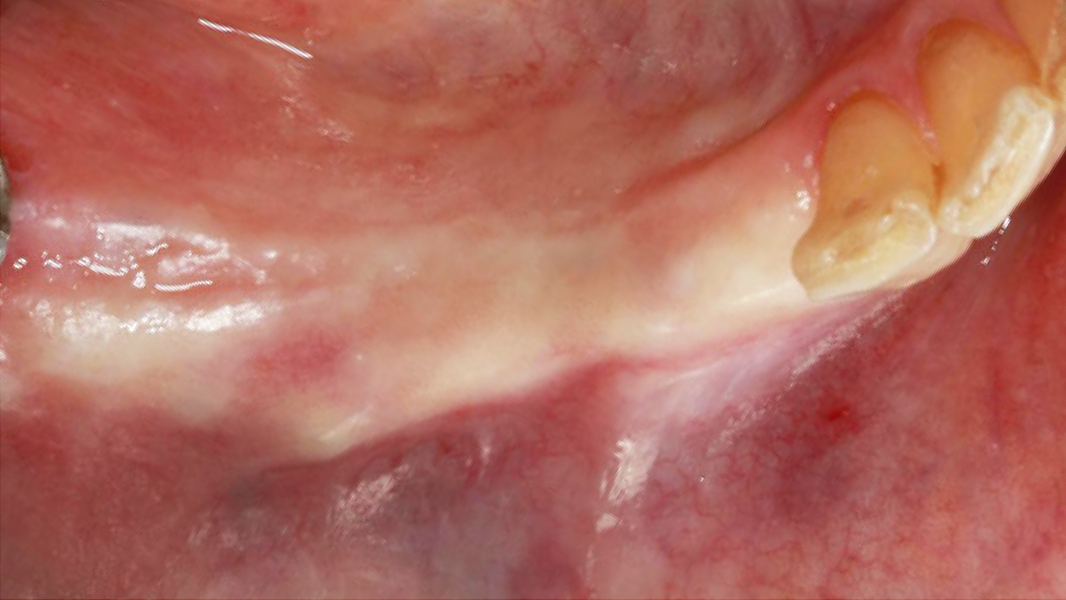
Rapid integration and natural remodeling
Product Features
- Allografts processed from voluntarily donated human tissues
- Advanced processing preserves the tissue integrity
- Natural bone composition – the native collagen is retained in the material6, 7
- High regenerative capacity and natural remodeling4
- Osteoconductive properties supported by a three-dimensional pore network and rough surface
- Shelf life: five years at ambient temperature (5–30°C)
Clinical Evidence
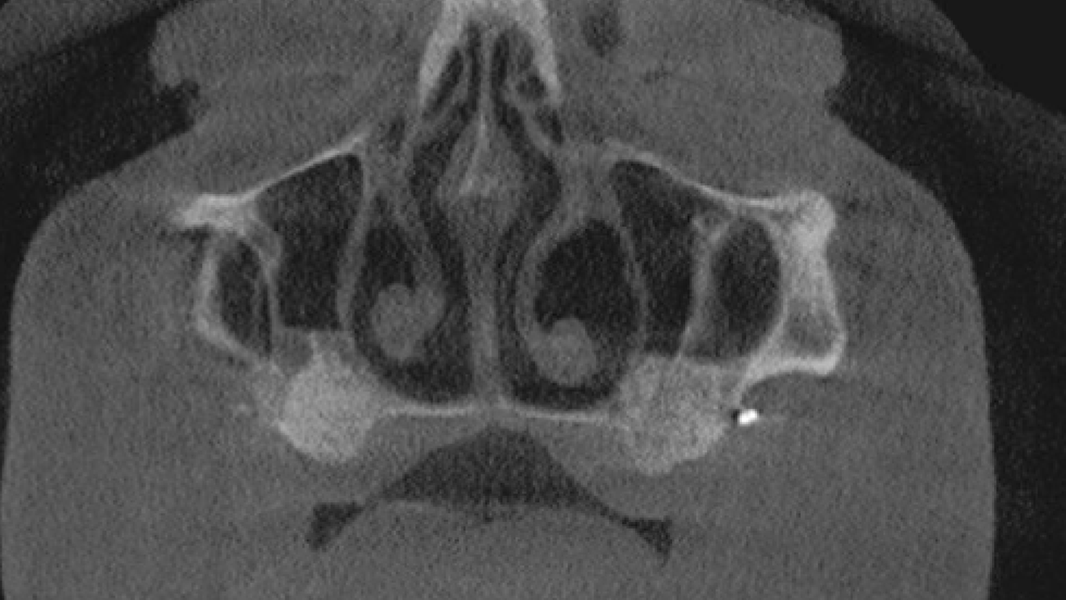
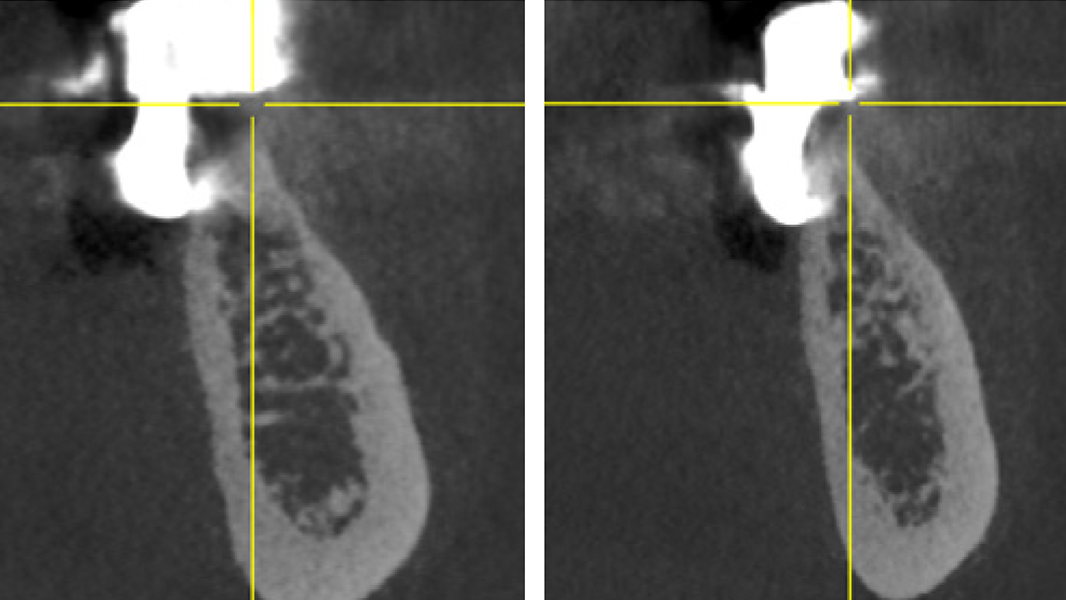
- A comparative clinical study demonstrated that MinerOss A produced significantly higher amounts of new bone in lateral sinus lift than bovine bone mineral.8
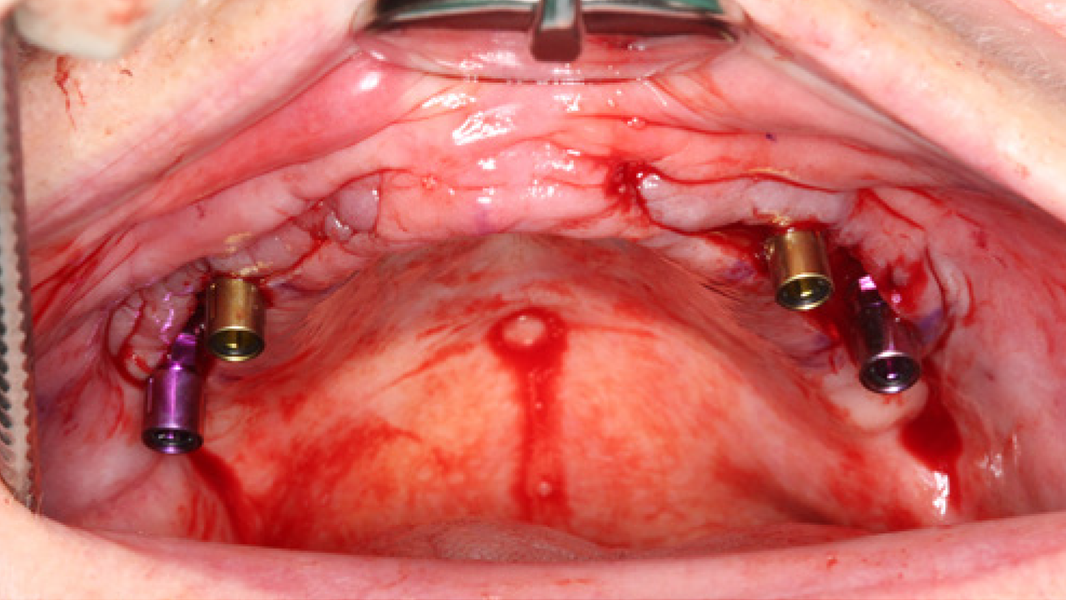
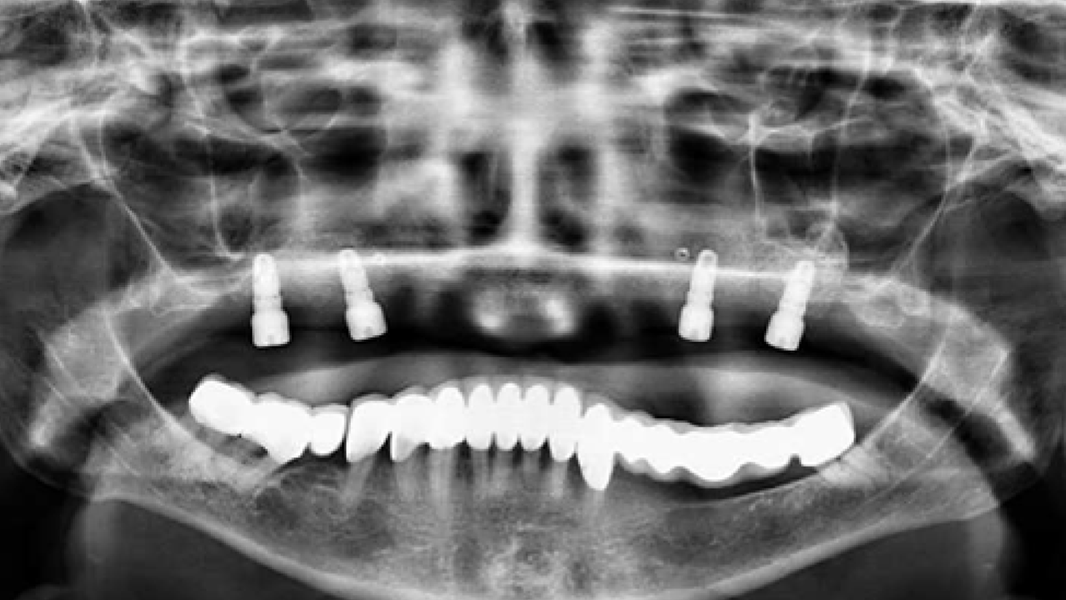
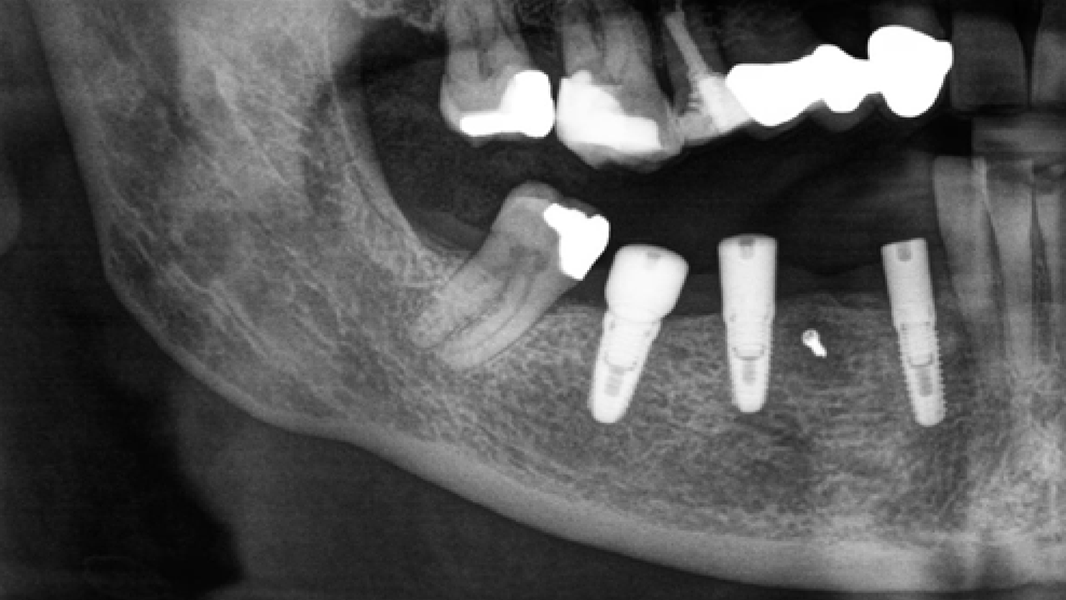
- The rapid turnover of MinerOss A shortens the reentry time and enables implant placement after 3–4 months.9, 10
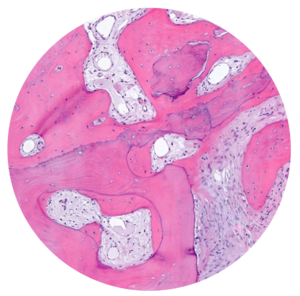
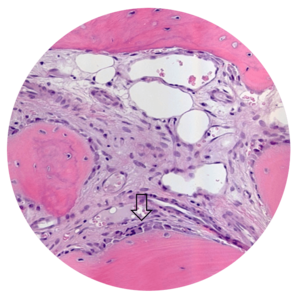
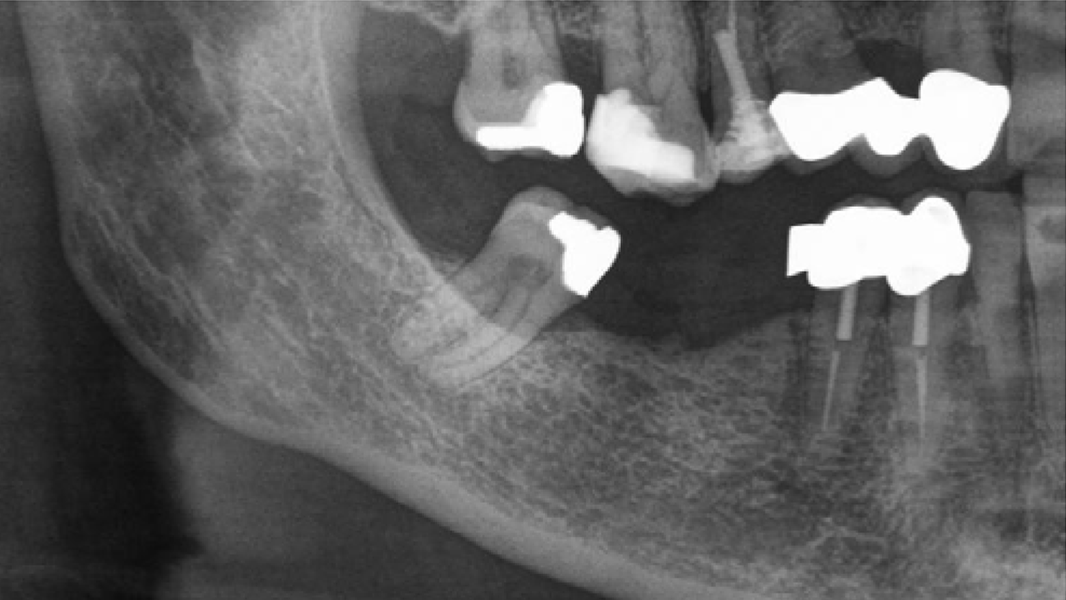
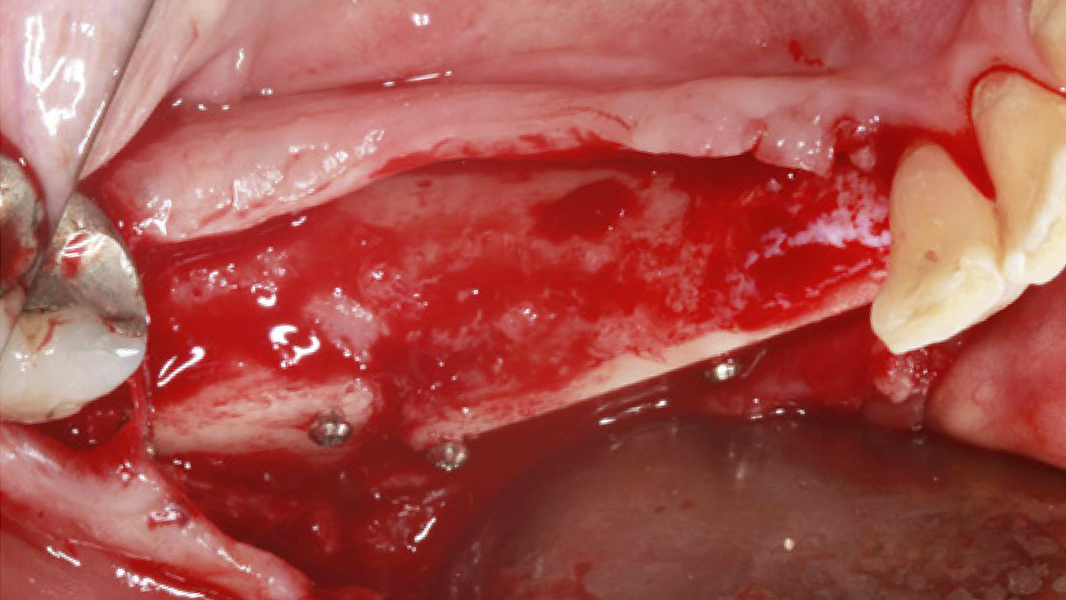
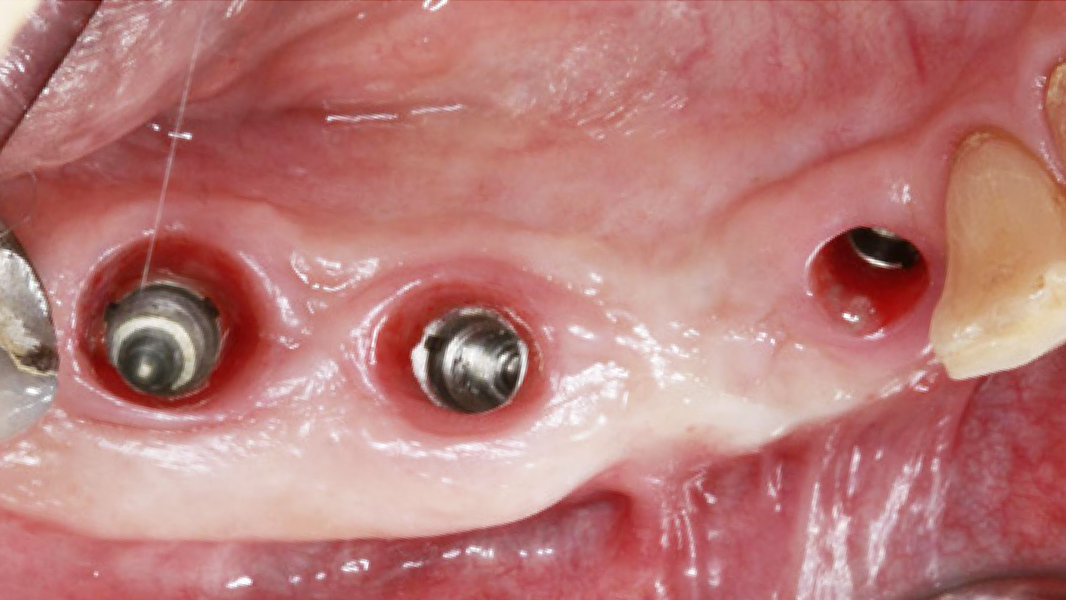
- MinerOss A completely remodels into patient’s bone; histological monitoring revealed well-vascularized lamellar bone without any avital remnants.3
- Using MinerOss A in bone augmentation procedures ensures excellent regenerative capacity4 and lower patient burden.11
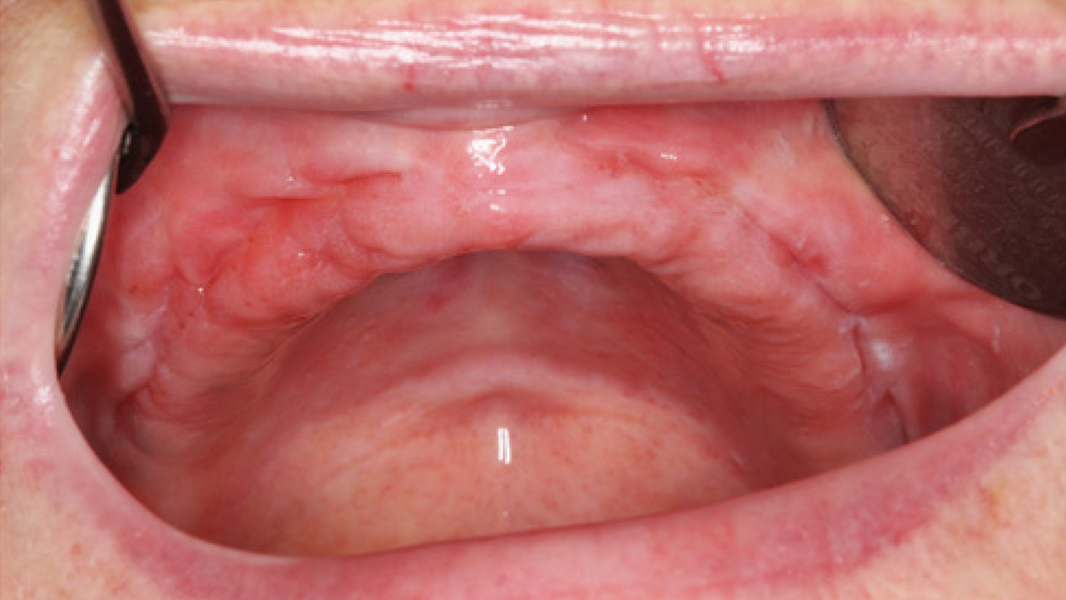
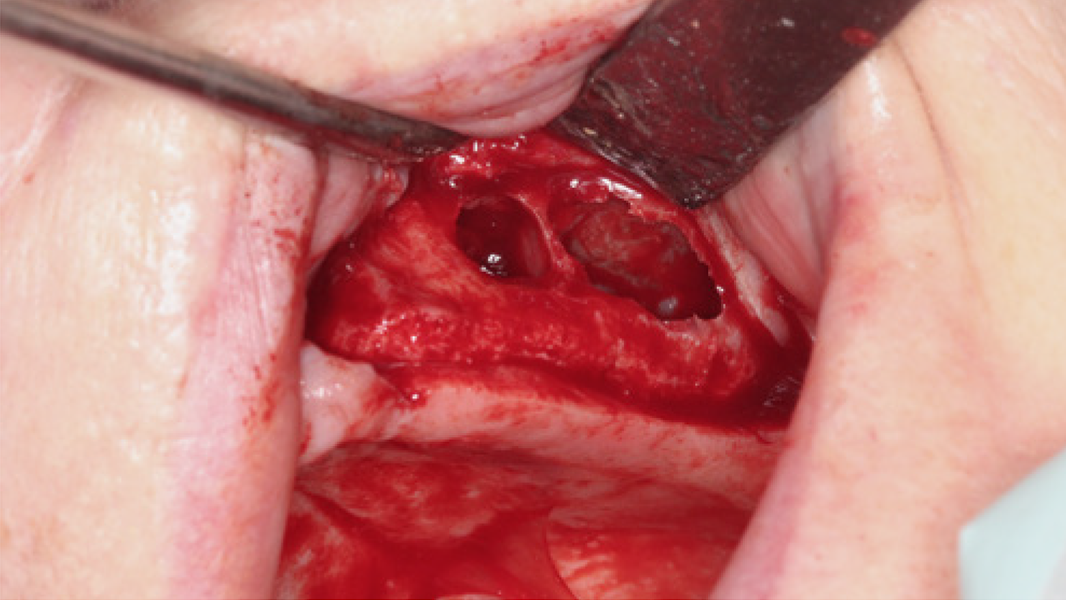
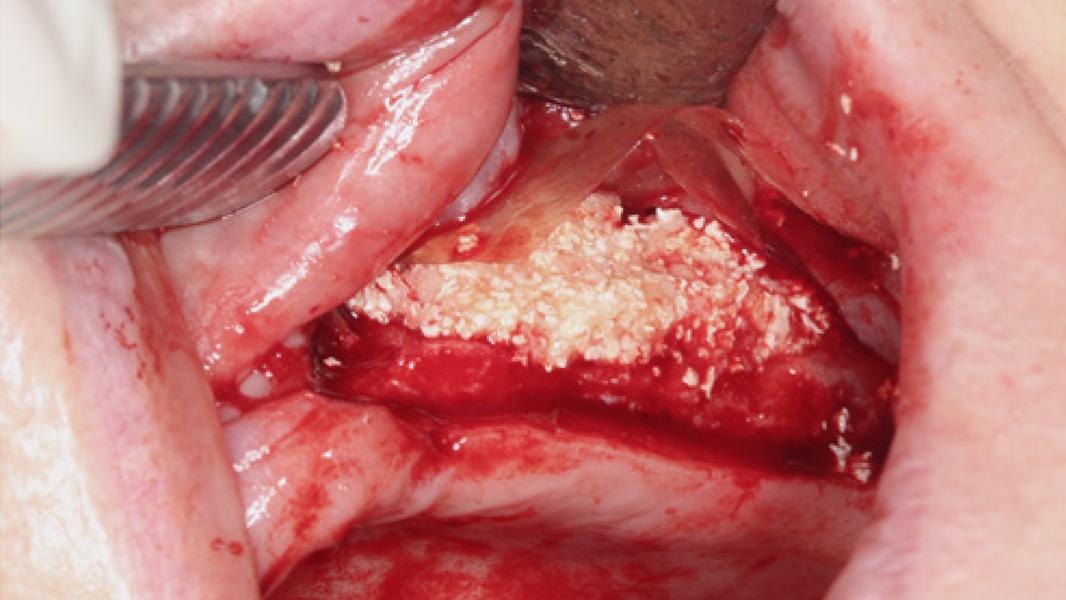
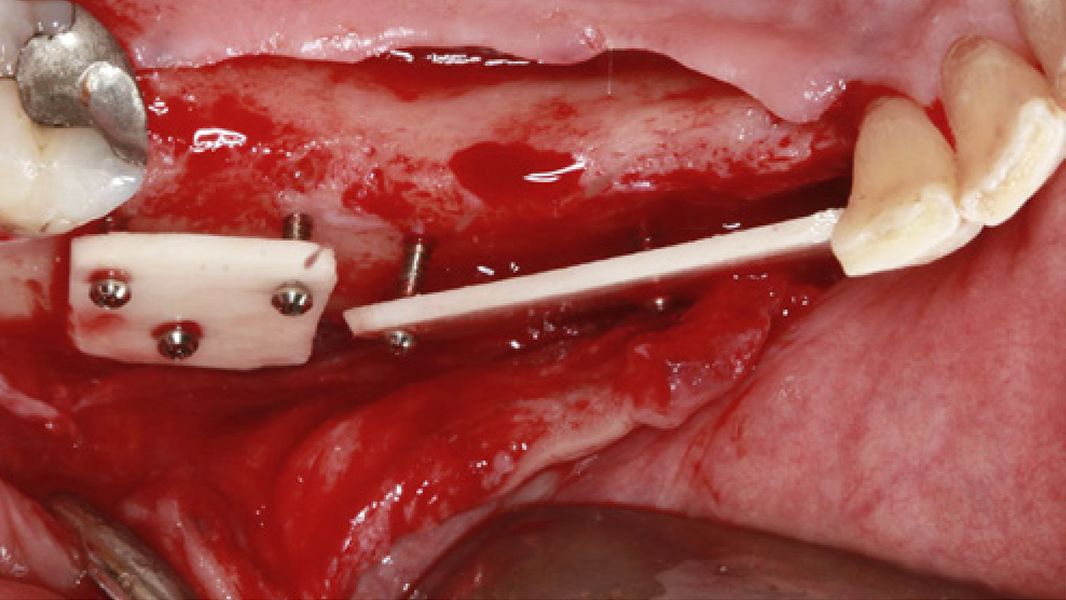
Regeneration and augmentation
- Preservation of extraction sockets
- Regeneration of missing bone tissue around dental implants
- Horizontal augmentation of the alveolar ridge
- Sinus augmentation
- 3D augmentations (horizontal and/or vertical) of the alveolar ridge
- Regeneration of periodontal osseous defects
- Regeneration after cysts and root tip resections
Allografts mixed with hyaluronic acid
Properties
Hyaluronic acid is a natural polysaccharide found in the extracellular matrix of various tissues. It plays a crucial role in maintaining tissue hydration and regulating cellular processes such as proliferation, differentiation, and signaling.12

MinerOss A + HYA product features
- Innovative combination
MinerOss A + HYA combines allogeneic bone granules with sodium hyaluronate (NaHyA) – a salt of hyaluronic acid.
- Exceptional fluid binding capabilities
Sodium hyaluronate exhibits exceptional fluid binding capabilities due to the numerous hydrophilic groups in its molecular structure, such as hydroxyl (-OH) and carboxylate (-COO-) groups.13
- Improved handling and precise application
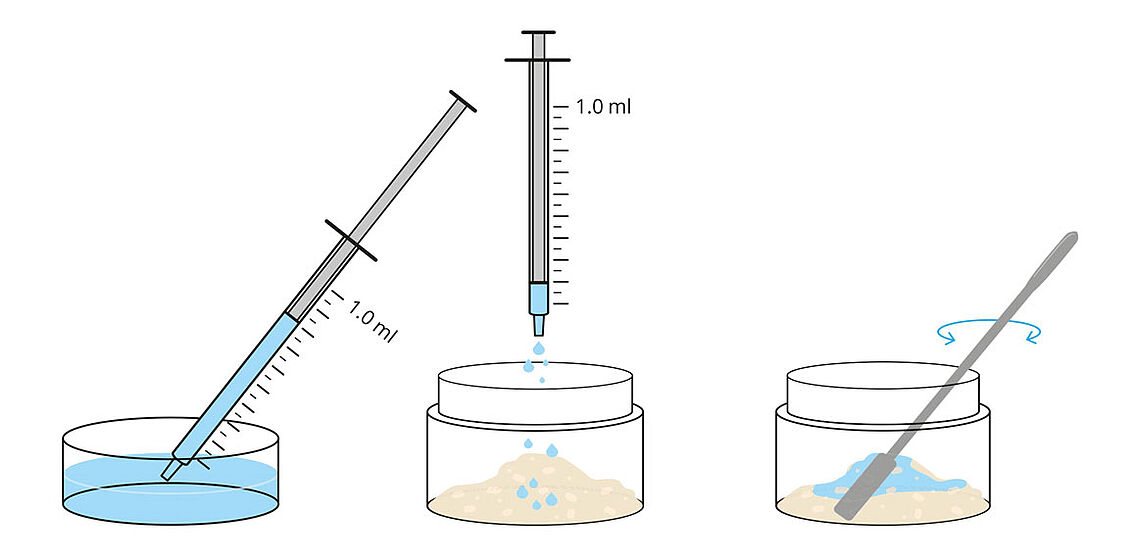
Sodium hyaluronate enables quick and easy preparation of the “sticky bone” upon hydration, thereby improving the handling and application of the bone substitute material.14, 15
- Unique composition
MinerOss A + HYA contains up to 20 times more hyaluronic acid than other commercial products. This results in better stickiness and malleability after hydration.16
Clinical Evidence MinerOss A + HYA
- 85 % of the surveyed clinicians rated the application of the sticky bone as “easier” or “much easie” compared to conventional particles.14, 17
- The preparation of the sticky bone was rated 8.9 on a scale of 1 (most difficult) to 10 (easiest).14
- On a scale of 1 to 10, clinicians rated the soft tissue healing within the first two weeks as 8.8.14, 17
The Allotec® process
High safety standards
After a thorough screening of donor history, a series of stringent serological tests (including exclusion of hepatitis B virus, hepatitis C virus, human immunodeficiency virus, and Treponema pallidum (syphilis)) combined with the Allotec purification process using C+TBA and gamma irradiation ensure maximum safety.
During the purification process, potentially present viruses are inactivated and bacteria destroyed. Furthermore, non-collagenous proteins are denatured, thus eliminating potential antigenicity.
High patient acceptance
Shorter treatment times
- Shorter surgery time: The ready-to-use allograft shortens surgery time by eliminating the need for a donor site.
- Lower patient morbidity and less pain for the patient: Avoiding a donor site eliminates the post-operative pain associated with a second procedure.
- Unlimited availability: It is predominantly derived from human donor femoral heads post hip replacement surgery.
- No clinical difference in final incorporation compared to autologous bone.3, 5, 18
References
1 Schmitt et al. Clin Oral Implants Res. 2013, 24, 576.
2 Solakoglu et al. Clin Implant Dent Relat Res. 2019, 21, 1002–1016.
3 Kloss et al. Clin Case Rep. 2020, 8(5):886-893.
4 Wen et al. J Periodontol. 2020. 91(2):215-222.
5 Kloss et al. Clin Oral Implants Res. 2018, 29, 1163.
6 Trajkovski et al. Materials 2018, 11(2):215.
7 Barbeck et al. Materials 2019, 12, 3234.
8 David Chávarri-Prado et al. Int J Periodontics Restorative Dent. 2024, 44(3):309-319.
9 Barone et al. Int J Periodontics Restorative Dent. 2024;44(6):685-696.
10 Kloss et al. Int J Implant Dent. 2024. 10(1):42.
11 Heimes et al. CIDRR. 2024. 26(1):170-182.
12 Garantziotis et al. Matrix Biol. 2019, 78-79:1-10.
13 Necas et al. Vet Med-Czech. 2008, 53(8):397-411.
14 Data on File. Limited Market Release. Users Feedback on Handling and Initial Healing Period 08/2023.
15 Data on File. Limited Market Release. Clinical Case Documentation.
16 Data on File. Marktforschung - Hyaluronsäure Hyaluronsäure-haltige Spritzen 01/2025.
17 Data on File. Entwicklungsbericht: Hyaluronat-Mix Evaluierung Produkteigenschaften 3: „Bone Paste-CTBA“. 03/2024.
18 Tunkel et al. Clin Case Rep. 2020. 9(2):947-959.